In our previous post, we explored the growing interest in linear peptides, peptidic and semi-peptidic macrocycles, as beyond-rule-of-5 modalities reshaping medicinal chemistry.
We also focused on SpiroChem’s capabilities in designing and synthesizing non-canonical amino acid analogues, key tools in the precise "molecular surgery" needed to optimize peptide-type hits and leads.
In this post, we turn to traditional and non-traditional dipeptide isosteres.
Traditional and non-traditional dipeptide isosteres are versatile building blocks that can be modularly inserted into lead sequences to address specific molecular liabilities.
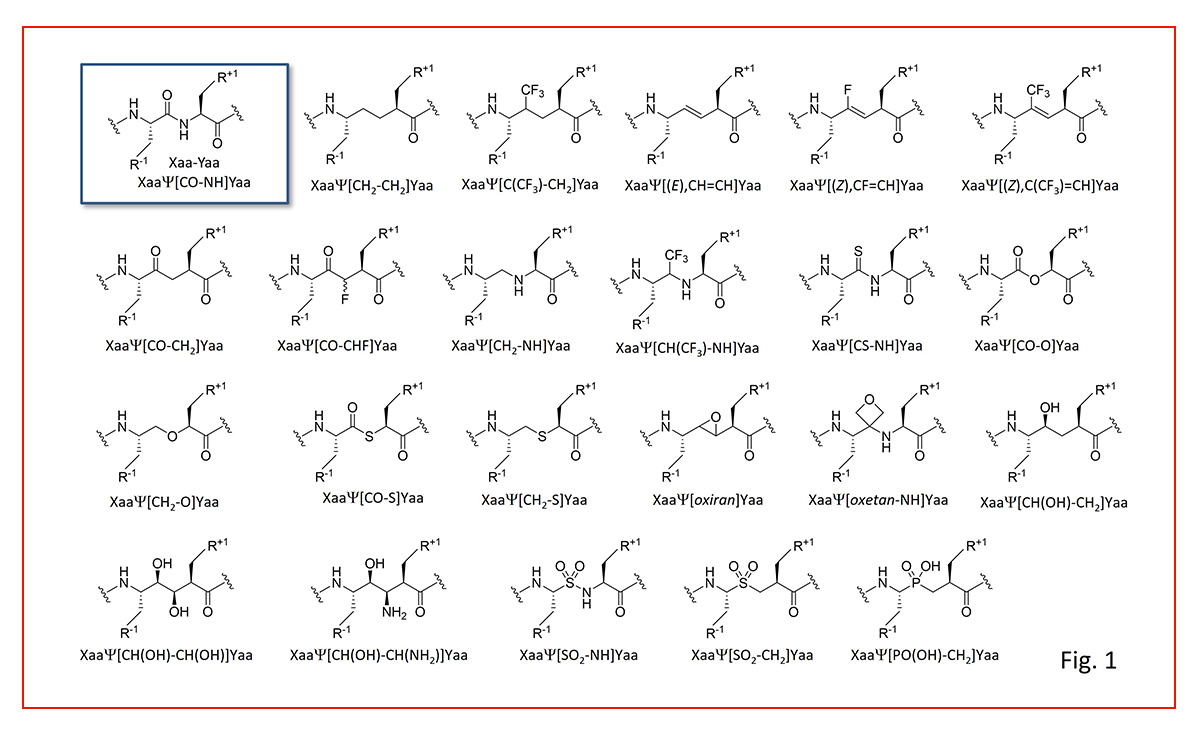
When it comes to replacing peptide bonds, either to block enzymatic hydrolysis or to mimic transition states of amide hydrolysis, we utilize a comprehensive toolbox of amide bond surrogates, including examples classified according to Spatola terminology (Fig. 1).
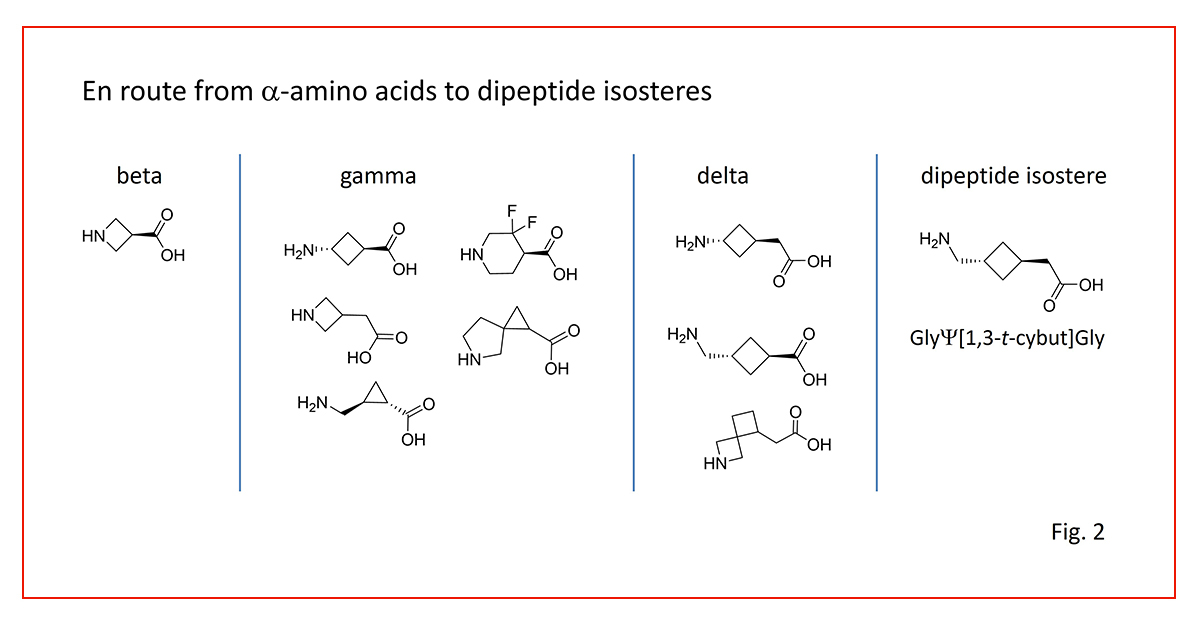
 Our conformational design strategy uses alpha-, beta-, gamma- and delta-amino acid homologues to fine-tune peptide backbone orientation, N-to-C isostere distances and overall shape (Fig. 2).
Our conformational design strategy uses alpha-, beta-, gamma- and delta-amino acid homologues to fine-tune peptide backbone orientation, N-to-C isostere distances and overall shape (Fig. 2).
 Atom count alone is not enough. Precise 3D architecture matters.
Atom count alone is not enough. Precise 3D architecture matters.
Detailed conformational design guidelines on how to mimic given C-alpha to C-alpha distances along a backbone are given in (Fig. 3).
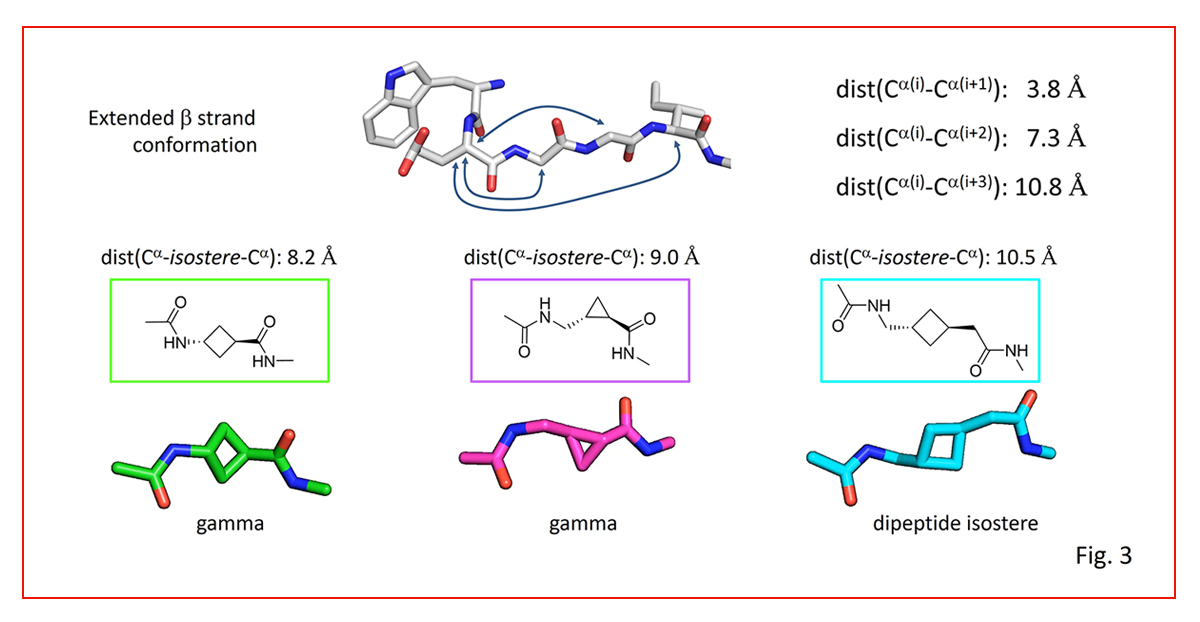 Peptidomimetics from our toolbox are showcased that enable small, controlled increases in inter-sidechain distances, crucial for modulating activity and selectivity.
Peptidomimetics from our toolbox are showcased that enable small, controlled increases in inter-sidechain distances, crucial for modulating activity and selectivity.
Aromatic elements can also help to increase conformational and metabolic stability.
Figure 4 compares an oxazole and a thiazole dipeptide isostere, highlighting how the topologically shorter thiazole surprisingly stabilizes a more extended conformation than the longer oxazole.
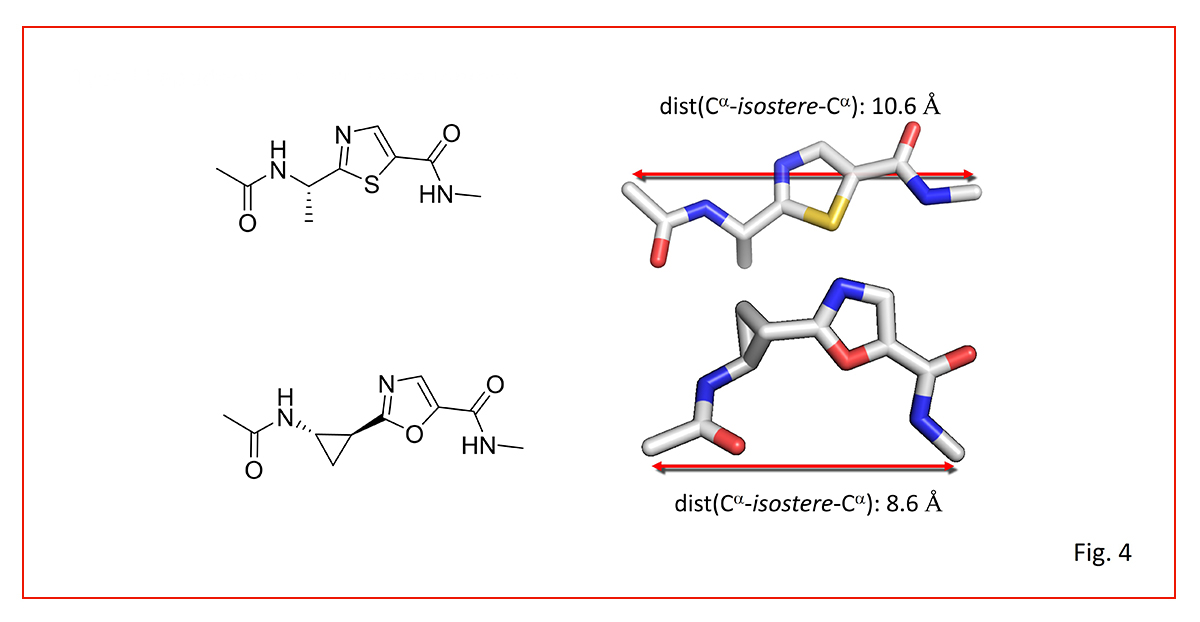 This is a case study on why detailed 3D conformational considerations provide sharper design guidelines than simple atom counts.
This is a case study on why detailed 3D conformational considerations provide sharper design guidelines than simple atom counts.
At SpiroChem, we support your peptide and macrocycle hit-to-lead and lead optimization campaigns through a hierarchical toolbox approach, blending cutting-edge molecular design, modern synthetic chemistry and solid-phase peptide expertise.

























